Differentiating patients with serious bowel disease from those with benign functional disorders, such as Irritable Bowel Syndrome (IBS), and minor colorectal disease such as haemorrhoids, hyperplastic polyps and simple Diverticular disease, can be very challenging since the symptoms are very common and overlap in these conditions.
To date clinicians have usually referred patients presenting with lower gastrointestinal symptoms for further investigation via colonoscopy, creating an escalating demand for these services. A colonoscopy is an invasive procedure with associated risks and it can be stressful and unpleasant for the patient. Colonoscopies tie up significant healthcare resources and are costly.
have usually referred patients presenting with lower gastrointestinal symptoms for further investigation via colonoscopy, creating an escalating demand for these services. A colonoscopy is an invasive procedure with associated risks and it can be stressful and unpleasant for the patient. Colonoscopies tie up significant healthcare resources and are costly.
Consequently endoscopy services are struggling to cope with increasing demand. In addition there is currently a low conversion rate to cancer in all symptomatic pathways i.e. colonoscopies are being conducted on a large numbers of patients who don’t have cancer.
NHS England spent approximately £178.4 million during 2014 on performing colonoscopies (based on NHS tariff price), yet with approximately 40% of those no pathologies were found. Identifying and prioritising those patients more likely to require urgent intervention could save significant costs, reduce waiting times and improve care.
‘There is evidence to suggest that triage using FIT at a cut-off around 10 μg Hb/g faeces has the potential to correctly rule out CRC and avoid colonoscopy in 75–80% of symptomatic patients. (Westwood et al. 1)’
In August 2023, The National Institute for Health and Care Excellence (NICE) updated the Diagnostic Guidelines for FIT, with DG30 being replaced by DG56.
The guideline highlights the diagnostic accuracy, clinical utility, and cost effectiveness of the HM-JACKarc, making it an ideal fit for the colorectal cancer referral pathway.
It is expected that this new guideline will:
- Reduce the number of colonoscopies performed
- Help triage patients to prioritise those most at risk of colorectal cancer
- Reduce waiting lists for colonoscopies
- Lead to improved consistency of best practice across the country
- Lead to better health outcomes and care experience
In July 2017 the National Institute for Health and Care Excellence published NICE Guidance DG30 “Quantitative faecal immunochemical tests to guide referral for colorectal cancer in primary care”
This focuses on the peer reviewed data for faecal immunochemical tests (FIT). Using this, the Diagnostics Advisory Committee created an economic model for each of the FIT methods under review. NICE DG30 recommends FIT for adoption in primary care to guide referral for suspected colorectal cancer in people without rectal bleeding who have unexplained symptoms but do not meet the criteria for a suspected cancer pathway referral outlined in NICE’s guideline on suspected cancer (recommendations 1.3.1 to 1.3.3).
It advises that results should be reported using a threshold of 10 micrograms of haemoglobin per gram of faeces to define the threshold for ruling out colorectal cancer. The ability to use a simple, easy to use, inexpensive diagnostic test will provide additional assistance in determining the appropriate patient pathway for further investigation.
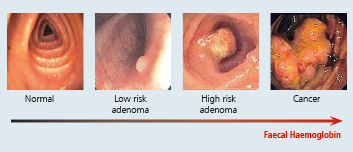
FIT may have a wider role as an objective triage tool in all symptomatic patients referred from primary care. The guidance also recognises that FIT detects a symptom of colorectal cancer (haemoglobin) that could also be associated with a range of other conditions. Data from studies reporting diagnostic accuracy for multiple target conditions in the same population, suggested that up to 28.9% of people with a false positive FIT result for colorectal cancer, did have some form of serious bowel pathology, such as inflammatory bowel disease or high risk adenoma.
The guidance concludes that it is plausible that the number of false-positive results that occur when using the tests to rule out colorectal cancer, could be partially offset by detecting other treatable bowel pathology.
 have usually referred patients presenting with lower gastrointestinal symptoms for further investigation via colonoscopy, creating an escalating demand for these services. A colonoscopy is an invasive procedure with associated risks and it can be stressful and unpleasant for the patient. Colonoscopies tie up significant healthcare resources and are costly.
have usually referred patients presenting with lower gastrointestinal symptoms for further investigation via colonoscopy, creating an escalating demand for these services. A colonoscopy is an invasive procedure with associated risks and it can be stressful and unpleasant for the patient. Colonoscopies tie up significant healthcare resources and are costly.
